We have reached a new phase in the things we will do with human life. It was reported by New Scientist last week that the world’s first “three parent baby” was born in Mexico.
The story of the family into which this child was born is a tragic one, and you cannot help but feel compassion for them. Married for almost 20 years, the unnamed couple tried to have children.
They suffered four miscarriages before giving birth to a baby girl. Unfortunately, the girl had inherited a rare genetic disorder from her mother and died at age six. A second daughter inherited the same disease and died after eight months.
The suffering of this family is untold.
But how does their suffering lead us to a three-parent baby? To understand this, we need a little bit of the scientific background.
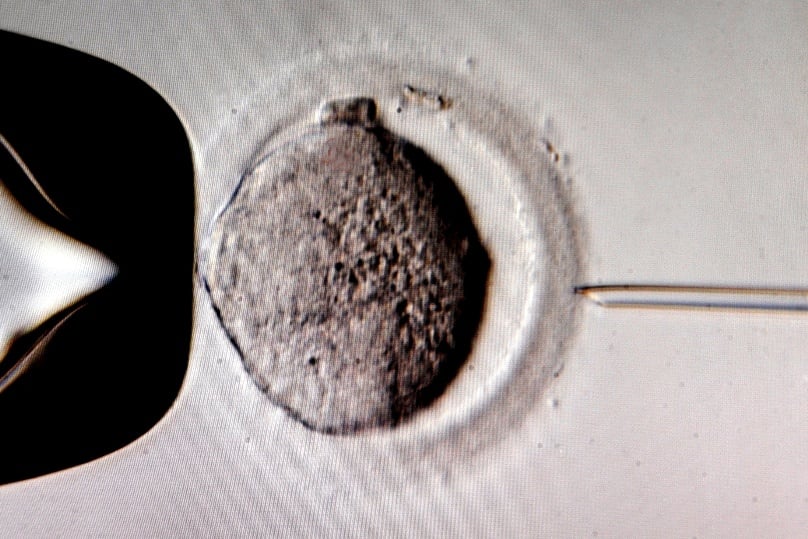
In most cases, the nucleus of an ovum contains 23 chromosomes before fertilisation and 46 chromosomes after fertilisation, which is usually what we think of when we talk about DNA. But in addition to the 46 chromosomes, there is a small amount of additional DNA contained in the mitochondria, which are located in the part of the ovum surrounding the nucleus. Problems in this “mitochondrial DNA” can cause certain genetic diseases.
There were two ways in which it is thought this can be overcome: pro-nuclear transfer and maternal spindle transfer.
Pro-nuclear transfer involves removing the nuclear material from a newly fertilised egg that has unhealthy mitochondria and transferring it into a donated “healthy” embryo which has had its own nuclear material removed. In this case, the second embryo is intentionally destroyed so that the other can be inserted. In every circumstance, it involves the taking of an unborn life. This method is currently legal in the United Kingdom but as far as I know, has not yet been used.
Maternal spindle transfer involves removing the maternal chromosomes prior to fertilisation and placing them into a healthy ovum which has had its chromosomes removed. This ovum is then fertilised. The difference here is that this process does not require the taking of a human life.
Because it was the more “ethical” approach, the couple chose maternal spindle transfer and while their opposition to taking human life is commendable, there are a few ethical considerations which they appear to not have sufficiently considered.
The creation of a three-parent baby still presents all of the ethical issues which are present in IVF, including the separation of the unitive and procreative aspects of the sexual act and the creation of more embryos than were planned on being implanted (in this case, five embryos were created and only one survived.)
The treating fertility doctor, Dr John Zhang, told New Scientist that “to save lives is the ethical thing to do”. However, Dr Zhang was not saving a life or curing a disease. He was just ensuring that a baby with a risk of a particular genetic disease was not born in the first place, making it less about saving lives and more about practising eugenics.
The next ethical issue overlooked is that there is no way of telling how having three genetic parents might affect future generations. Historical methods of trying to treat mitochondrial disease, including injecting mitochondria from another woman into the ovum, resulted in babies being born with a greater disposition to other genetic diseases. Only time will tell if this technique also creates future physical risk to the baby.
In addition to the unknown physical impacts, the effects on the child’s understanding of his own identity is equally unchartered territory, as are the legal implications because there are now three people who validly have a legal “claim” on the baby.
And if we are being blunt, having a “claim” on the baby is the reason why we are in this ethical minefield in the first place. The couple could have easily used a donor egg to avoid the potential genetic disease (noting this still carries the ethical problems of IVF), but if they had done that, the child would not be genetically related to the intended mother. The reason this new technology was sought is because the intended mother desired a biological connection to the child. As understandable as that yearning is, and as awful as the tragedies these parents have had to endure, it does not justify the prioritisation of their own desires over potential risks to the child.
It is also important to consider the motivations of the fertility industry in this. UK researchers estimated that 10-20 families per year might use the technology, indicating a very small demand for this technology from people with mitochondrial disease. If they were the only people to whom this service could be marketed, the business would not be lucrative enough to justify the funds spent on perfecting the technology.
But I’m guessing the fertility experts see the potential for a wider market: the same technology can be used by a lesbian couple to have a child which is biologically related to both of them and it is this market which will expand in years to come, particularly if more countries legalise same-sex marriage. The impact on a generation yet unborn is another one of the unintended consequences of changing the marriage law.
